Nutrition science has long circled around a deceptively simple question: Can we meaningfully shift the human microbiome through diet alone? A major Stanford study, published in Cell, has now given one of the clearest answers to date—and it’s a finding that challenges long‑held assumptions about fibre, fermented foods, and the way modern guts respond to dietary change.
The trial, which placed healthy adults on either a high‑fibre or high‑fermented‑food diet for ten weeks, set out to test two of the most widely promoted gut‑health strategies. Both are staples of whole‑food living. Both are backed by decades of mechanistic research. But the study found that they do very different things inside the body—and only one produced a broad, measurable reduction in inflammation.
That’s important because chronic low‑grade inflammation is the quiet engine behind many of today’s non‑communicable diseases.
The study
Researchers recruited 36 adults and randomised them into two groups:
- High‑fibre diet: participants doubled their fibre intake to around 45 g/day, focusing on legumes, whole grains, nuts, seeds, fruits, and vegetables.
- High‑fermented‑food diet: participants consumed around six daily servings of foods like yoghurt, kefir, kimchi, sauerkraut, kombucha, and fermented brine drinks.
Across 17 weeks, the team collected stool, blood, and immune‑profiling data using some of the most advanced tools available—metagenomics, proteomics, metabolomics, and deep immune phenotyping. The result was one of the most detailed maps ever created of how diet interacts with the microbiome–immune axis.
Surprise finding
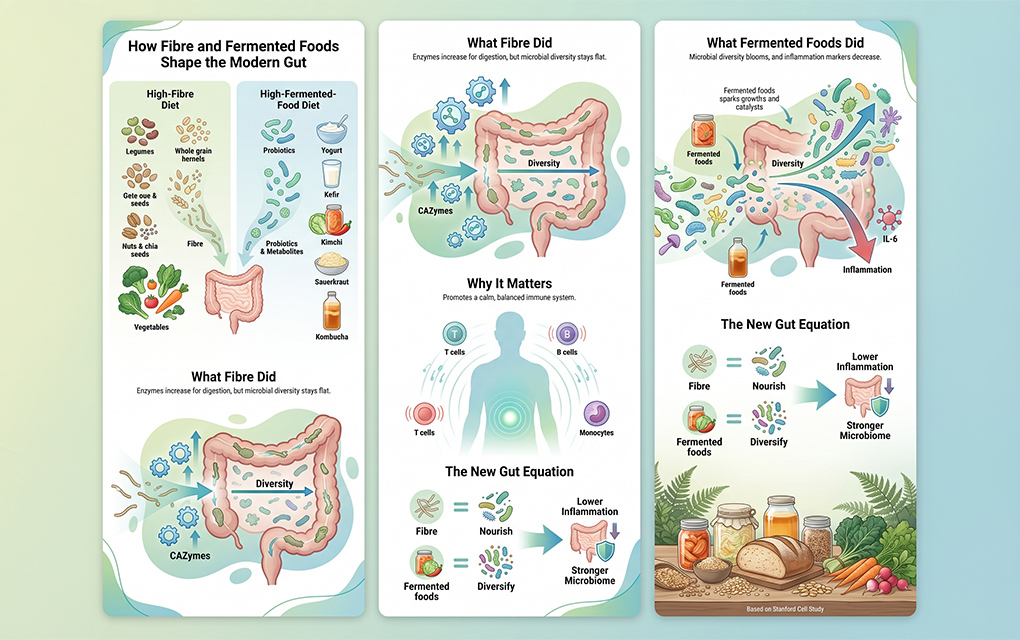
Fibre has long been championed as the cornerstone of gut health. It feeds beneficial bacteria, increases short‑chain fatty acid production, and supports metabolic resilience. All of that remains true. But the Stanford team found something surprising:
1. Fibre improved microbial function, not diversity.
Participants eating more fibre showed a significant rise in carbohydrate‑active enzymes (CAZymes)—the molecular machinery microbes use to break down plant fibres. Their microbiota became better at digesting fibre, even though the overall species diversity did not increase.
This suggests fibre strengthens the ecosystem you already have.
2. Many participants couldn’t fully ferment the extra fibre.
Unprocessed carbohydrates began appearing in stool samples, indicating that the microbiome of many participants—typical of industrialised populations—lacked the fermentative capacity to keep up with the increased load.
This aligns with research showing that Westernised microbiomes have lost fibre‑degrading species over generations.
3. Immune responses varied dramatically.
Some participants showed reduced inflammatory signalling; others showed increases. The strongest predictor of who benefited? Baseline microbiome diversity. Those starting with richer microbial ecosystems responded more favourably.
This reinforces a key insight: fibre works best when the gut is already diverse enough to use it.
Fermented foods
If fibre strengthened the existing ecosystem, fermented foods did something more dramatic.
1. Fermented foods increased microbiome diversity—cohort‑wide.
Across the group, microbial diversity rose steadily throughout the intervention and continued rising even during the “choice” phase when intake dropped. Diversity is one of the strongest known markers of gut resilience.
Crucially, the new species detected were not primarily the microbes found in the fermented foods themselves. Instead, fermented foods appeared to make the gut more hospitable—more permeable to environmental microbes or more supportive of previously undetectable strains.
2. Inflammation dropped—significantly and broadly.
Nineteen inflammatory markers decreased, including IL‑6, IL‑10, and IL‑12b. IL‑6 is especially important: it spikes in severe infections, rises in rheumatoid arthritis, and is elevated in depression. It is not a “gut molecule”—it is a whole‑body alarm signal.
This finding has captured the attention of clinicians and metabolic health advocates alike. As Dr David Reiner noted in a LinkedIn post, IL‑6 is often elevated long before symptoms become diagnosable. A quiet, chronic elevation can manifest as fatigue, brain fog, or a persistent sense of being “off”—the very symptoms many people struggle to explain.
3. Immune activation decreased across multiple cell types.
Participants showed reduced signalling in T cells, B cells, and monocytes—suggesting a systemic calming of the immune system.
This is one of the clearest demonstrations to date that fermented foods can modulate inflammation at a population level.
Headline finding
The trial’s headline finding—fermented foods outperform fibre in reducing inflammation—has sparked lively debate. But the nuance matters.
As several nutrition experts in the LinkedIn thread pointed out, these are not competing strategies. They are complementary. Fibre nourishes the microbes you already have. Fermented foods appear to shift the ecosystem itself.
Modern guts may lack the microbial “starter kit” needed to fully benefit from fibre.
Industrialised lifestyles—sanitised environments, low‑fibre diets, reduced microbial sharing—have eroded microbial diversity. Without a diverse baseline, simply adding more fibre may not be enough to rebuild what’s missing.
Ecosystem catalysts
Even if the microbes in fermented foods don’t permanently colonise the gut, their presence—and the metabolites they produce—may create conditions that allow dormant or low‑abundance species to flourish.
This is consistent with the study’s finding that diversity gains were not directly tied to the microbes in the foods themselves.
For Whole Food Living readers—many of whom already eat plant‑rich, minimally processed diets—the study offers a valuable reframing.
1. If you already eat plenty of fibre, fermented foods may be the missing piece.
Many people who “eat well” still experience low‑grade symptoms: sluggishness, fogginess, irregular digestion. The Stanford trial suggests that without microbial diversity, fibre alone may not shift the system.
2. Diversity is a health marker worth protecting.
Low microbial diversity is linked to obesity, diabetes, autoimmune conditions, and inflammatory disorders. Increasing diversity through fermented foods may be one of the most accessible tools we have.
3. The benefits may require ongoing intake.
As several experts noted in the LinkedIn discussion, the durability of diversity gains is still unknown. Early evidence suggests benefits fade when intake stops—much like exercise, sleep, or any other adaptive system.
4. Not all fermented foods are equal.
The strongest correlations in the study were with yoghurt and fermented brine drinks. But traditional ferments—kimchi, sauerkraut, kefir—remain rich sources of microbial metabolites and bioactive compounds.
A new framework
The emerging consensus from researchers and clinicians is not that “fermented foods beat fibre.” It’s this: A resilient microbiome needs both nourishment and new recruits.
- Fibre strengthens the existing community.
- Fermented foods expand it.
- Together, they create a more adaptable, less inflammatory ecosystem.
For New Zealanders—where traditional Māori fermentation practices, Pacific food cultures, and modern whole‑food movements intersect—this synergy offers a powerful, culturally resonant path forward.
The same applies across Asia, where long‑standing traditions of kimchi, natto, tempeh, dosa batter, and medicinal broths are being re‑examined through the lens of microbiome science and metabolic health.
In Europe, centuries‑old practices such as sourdough, kefir, sauerkraut, and regional fermented cheeses are being rediscovered as part of a broader shift toward sustainable, minimally processed diets rooted in local identity.
And in North America, where Indigenous foodways, immigrant culinary traditions, and contemporary wellness movements converge, a growing emphasis on fibre‑rich, plant‑forward eating is reshaping conversations about health, equity, and the future of food.
Where to next?
The Stanford team emphasises that this was a small study in healthy adults. Larger trials, longer follow‑ups, and studies in people with inflammatory or metabolic conditions are needed. But the implications are already reshaping how clinicians think about gut‑directed nutrition.
If future research confirms these findings, fermented foods may become a formal part of dietary guidelines—not just for gut health, but for systemic inflammation.
And fibre? It remains essential. But its full power may best be unlocked only in a microbiome with the diversity to use it.





